Spectrophotometric Determination of Amodiaquine in Amodiaquine-containing Tablets
DOI:
https://doi.org/10.4314/Palavras-chave:
amodiaquine, ultraviolet spectrophotometry, potencyResumo
Background: Amodiaquine (AQ) is a medication on the World Health Organisation's List of Essential Medicines and is recommended for the treatment of malaria in combination with artesunate (ASAQ) or sulphadoxine-pyrimethamine (SPAQ) to reduce the risk of resistance. This study evaluated the amodiaquine potency of amodiaquine-containing formulations.
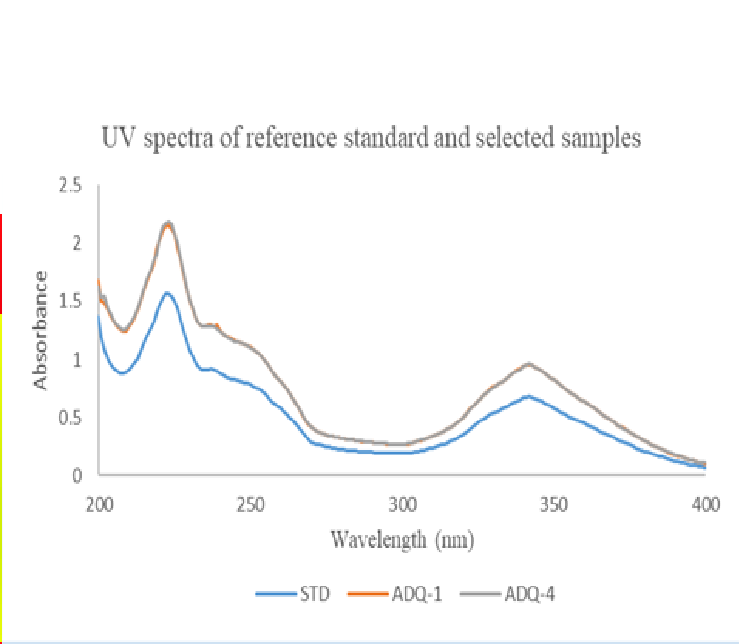
Methods: Identity and potency were established using amodiaquine dihydrochloride dihydrate analytical reference standard. The amodiaquine content of each sample was measured by assessing its absorbance after dissolution and proper dilution in 0.1N HCl using ultraviolet (UV) spectrophotometry (USP Monograph).
Results: A total of ten co-blistered and fixed-dose combination amodiaquine-containing tablets were randomly collected and tested. The mean percentage amodiaquine content of the tablets was determined from the absorbance values at 223, 237 and 342 nm. All the samples were found to contain amodiaquine as one of the active pharmaceutical ingredients of the tablets. Validation tests indicate excellent linearity (correlation coefficient, r > 0.999), good precision (relative standard deviation (%), < 2), and accuracy [recovery (%) for AQ (100.16, 99.89 and 100.06); ASAQ (100.14, 100.12 and 100.14) and SPAQ (99.16, 100.11 and 99.21) at 223, 237 and 342 nm, respectively], of the method. The active amodiaquine % contents of the samples were evaluated and found to be in the range of 101.58 – 106.95%.
Conclusion: All the samples passed the identification and potency tests based on the USP acceptance criteria of 93.0 – 107.0% and are of good quality with respect to the active amodiaquine content of the formulations.
Referências
Downloads
Publicado
Edição
Seção
Licença
Direitos autorais (c) 2026 Nigerian Journal of Pharmaceutical Research

Este trabalho está licenciado sob uma licença Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
