Development of an Ion-Activated In Situ Gel System for Enhanced Ocular Delivery of Timolol Maleate
DOI:
https://doi.org/10.4314/Palabras clave:
ion-sensitive, in situ gel, glaucoma, timolol maleate, sustained releaseResumen
Background: Glaucoma is a chronic eye disease that requires twice daily administration of eye drops. This study aims to develop a novel ion-activated in situ gel (ISG) formulation for once daily ocular administration in the management of glaucoma.
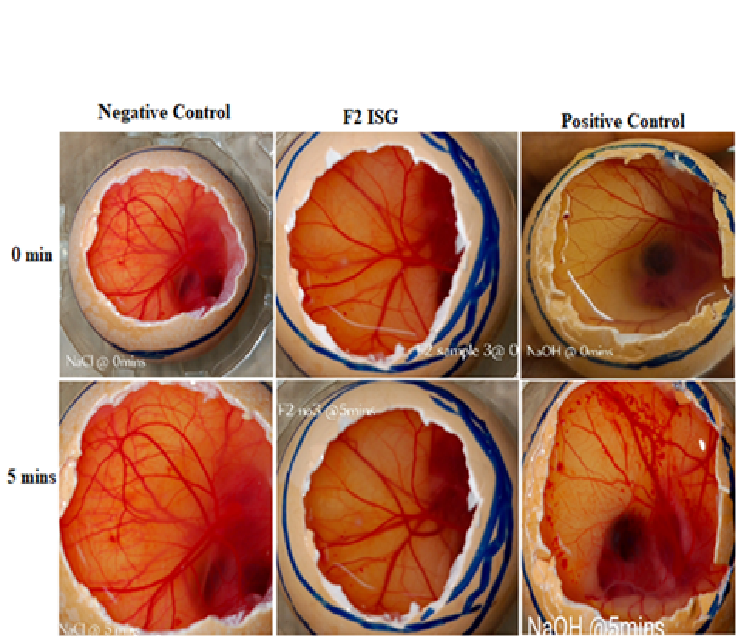
Materials and Method: Timolol maleate (TM) in situ gels, F1 to F12 were formulated using different concentrations and combinations of gellan gum (GG), sodium alginate (SA) and thermosensitive polymers of varying viscosities. The formulations were evaluated for physicochemical characteristics, gelling capacity, gelling temperature, drug content, rheological behavior, release kinetics, toxicity and irritability using the hen’s egg test- chorioallantoic membrane (HETCAM) assay.
Results: All formulated ISGs were clear, colorless and free of particles. The pH of all formulations was between 5.27 and 6.81 which is within the acceptable range for comfort. The viscosity of the formulations at a shear rate of 100/s showed F1 (GG 0.2%, SA 0.5% w/v) had the lowest viscosity of 2 cp and F9 (GG 0.2% w/v, HPMC MV 0.6% w/v), had the highest viscosity of 202 cp at room temperature. Drug release studies showed sustained release of approximately 50% of the ISGs in 3 h compared to 30 mins for conventional TM solution.
Conclusion: ISGs formulated with the right combination of ion-sensitive polymers can potentially increase the residence time of TM in the eye and improve patient adherence to therapy.
Referencias
Descargas
Publicado
Número
Sección
Licencia
Derechos de autor 2026 Nigerian Journal of Pharmaceutical Research

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-SinDerivadas 4.0.
