Behavioural, Sedative and Anticonvulsant Activities of Ethanol Extract of the Leaf of Lagenaria breviflora (Benth) in Mice
DOI:
https://doi.org/10.4314/njpr.v19i1.8کلمات کلیدی:
Lagenaria breviflora, LD50, Novelty-induced behaviour, Anticonvulsant, Maximal electroshock, Pentylenetetrazoleچکیده
Background: Lagenaria breviflora Benth is used in West Africa as a traditional remedy for the treatment of gastrointestinal disorders, the treatment of human measles, digestive disorders, and as a wound antiseptic.
Objectives: This study investigated the behavioural, sedative, and anticonvulsant activities of the ethanol extract of Lagenaria breviflora (EOLB) in mice, and its possible mechanism(s) of action.
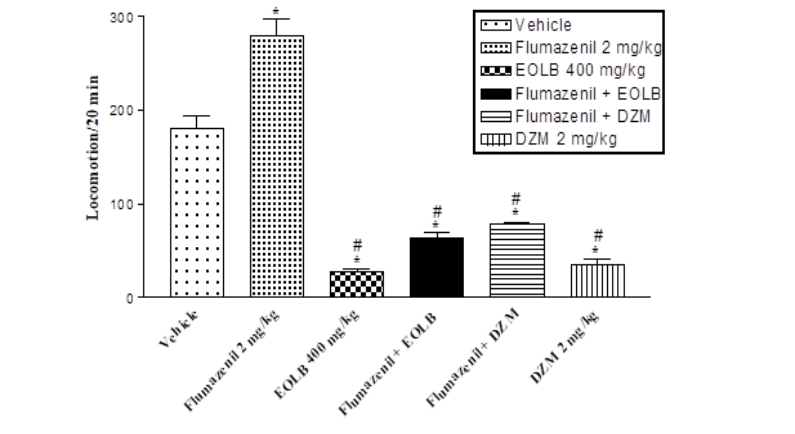
Materials and Methods: The CNS activities of EOLB (100, 200, and 400 mg/kg, i.p., n=6) were evaluated using animal models of novelty-induced behaviours (NIB), sedation (ketamine-induced hypnosis), and anticonvulsion (maximal electroshock (MES)), pentylenetetrazole (PTZ), and strychnine). Probable mechanism(s) were evaluated using an antagonist (flumazenil, 2 mg/ml, i.p.) and an agonist (amphetamine (2 mg/kg, i.p.) on NIB.
Results: LD50 values obtained were 1225 mg/kg and greater than 5000 mg/kg for the intraperitoneal and oral routes respectively. The EOLB at all doses tested significantly (p<0.01) inhibited the NIB. The EOLB (400 mg/kg) significantly (p<0.01) shortened sleep latency and significantly (p<0.05) prolonged the total sleeping time induced by ketamine (100 mg/kg, i.p.). EOLB at 400 mg/kg gave 33.3 % protection against HLTE caused by the MES-induced convulsion, significantly (p<0.01) prolonged the time of death, and offered 83.3 % protection against convulsion caused by PTZ. Flumazenil and amphetamine (2 mg/kg, i.p. respectively) significantly (p<0.001) reversed the effects of the EOLB on NIB.
Conclusions: The study concluded that the ethanol leaf extract of Lagenaria breviflora is safe orally and mildly toxic intraperitoneally. The extract showed significant depressive activity on the central nervous system, sedative and anticonvulsant activities which may be mediated through augmentation of GABAergic or inhibition of the dopaminergic neural pathways, thus supporting some of the tradomedicinal claims of the uses of Lagenaria breviflora.
مراجع
دانلودها
چاپ شده
شماره
نوع مقاله
مجوز
حق نشر 2023 Nigerian Journal of Pharmaceutical Research

این پروژه تحت مجوز بین المللی Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 می باشد.
